Benzodiazepines are a widely prescribed group of medicines with a range of clinical uses that include treating anxiety, insomnia, acute muscle spasms and managing alcohol withdrawal.However, this group of medicines are often used outside of prescribing recommendations. They can be used by people, including those accessing addiction and homeless services, as part of a poly drug use culture in Ireland. Like all medicines, benzodiazepines can produce side effects. They are also dangerous if mixed with other substances, particularly depressant type drugs such as opioids or alcohol.
Over the last decade, of concern is the emergence of ‘new’ benzodiazepines with an increase in the number, type and availability. The EU Early Warning System has detected an increasing number of ‘new’ benzodiazepines available on Europe’s drug market. The European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) is currently monitoring 28 new benzodiazepines. Some 23 of these have been first detected in Europe over the last 5 years. Some of the benzodiazepines new to the EU market may be called ‘designer’ or ‘novel’. Some may have been approved for use in some countries in the past, others may be found in patent literature but were never been brought to market, whereas some are novel compounds produced in illicit manufacturing plants often in the Far East.
Source: The European Monitoring Centre for Drugs and Drug Addiction, 2019
Risks
NPS benzodiazepines appearing on the market are of concern due to high potency at low doses. This can produce strong sedation, respiratory depression and amnesia. Additional adverse effects include loss of coordination, drowsiness, dizziness, blurred vision, slurred speech, and, in some cases, death. Risks are increased if one or more substances are used by the person, or if tablets are pressed to contain one or more substance.
Four drugs — etizolam, diclazepam, flubromazolam and phenazepam — account for over 80% of all tablets containing new benzodiazepines that have been seized in Europe since 2005.
In 2016, over half a million tablets containing new benzodiazepines, or similar substances, were seized, some two-thirds up on the number seized in 2015.
UK Advisory Council on the Misuse of Drugs (ACMD) ‘Novel Benzodiazepines, A review of the evidence of use and harms of Novel Benzodiazepines
In April 2020, the ACMD published a new report in relation to the harms caused by Novel Benzodiazepines in the UK, with a particular focus on 13 benzodiazepines that have been notified to the EMCDDA but are currently not classified under the Misuse of Drugs Act. The Report concludes that there is no evidence for legitimate medicinal uses of these substances in the UK.
There is evidence from drug seizure data of prevalence for 3 of the 13 compounds in the UK, namely flualprazolam, flunitrazolam and norfludiazepam, highlighting their availability and harm.
As cited in the Report, in the period July to September 2019, benzodiazepines were the most commonly identified substances by WEDINOS (a Welsh drug checking organisation). Between October and December 2019, flualprazolam was the 5th most commonly identified substance. Over the course of 2019, the presence of flualprazolam in WEDINOS samples increased significantly, with identifications almost doubling from 17 in the second quarter, to 30 in the third quarter of 2019. In many cases, these samples were counterfeit diazepam tablets or white ‘Xanax’ bars. This is of concern because of the confirmed potency of this compound and its links to deaths across Europe.
The National Programme on Substance Abuse Deaths for England and Wales reported 3 drug-related deaths where flualprazolam was specifically stated in the cause of death or implied cause of death due to multiple drug toxicities. A further 2 cases were reported where flualprazolam was detected at post mortem.
In Scotland, 7 flualprazolam-related deaths had been reported to Record of Scotland (data up to March 2020). In all 7 cases, the death was not caused by flualprazolam alone: it was just one of (usually) several substances implicated in the cause of death, in combination.
New Combinations of Benzodiazepines Appearing on the Irish market
The potential for diverting licit prescription supplies to the illicit market has reduced due to tightening of prescribing practices but of concern is the growing availability of these ‘new’ benzodiazepine type substances on the market in Ireland. There is evidence in Scotland, where ‘street’ or unlicensed benzodiazepines were involved in 85% of the 792 deaths in 2018 where a benzodiazepine was implicated, while medicinal ‘prescribed’ benzodiazepines were reported in only 30%.
In Ireland, illicitly sources tablets are called ‘street tablets’ with newer varieties titled ‘fake’. The most well know being ‘benzo/Xanax sticks’ or 'bars' that have been on sale for the past number of years.
Through Forensic Science Ireland and the HSE National Drug Treatment Centre, we are aware of some of the novel substances that have been emerging in Ireland for the past number of years.
In 2016 the Forensic Science Ireland laboratory detected new benzodiazepines such as phenazepam, nitrazolam, etizolam and chlorodiazepam in drug seizures.
In order to monitor the profile of benzodiazepines being used in the addiction population and to determine if any new benzodiazepines are being used, a multi-residue method was developed by the HSE National Drug Treatment Centre to monitor trends.
A total of 200 samples were analysed by LC-MS. Positives were as follows:
(86.5%) oxazepam, (84.5%) temazepam, (79.5%) nordiazepam, (12.%) 2- hydroxyethylflurazepam, (66.5%) alprazolam, (64.4%) α -hydroxyalprazolam, (8%) lorazepam, (7%) lormetazepam, (4%) flurazepam, (3.5%) etizolam, (0.5%) bromazepam, and (0.5%) 7-aminoclonazepam. Lorazepam could be present from intake of Lorazepam and/or Lormetazepam or Diclazepam. One patient was positive for six benzodiazepines or metabolites: nordiazepam, temazepam, oxazepam, alprazolam, and α-hydroxyalprazolam and etizolam, probably indicating diazepam, alprazolam and etizolam use.
There have been 2 flualprazolam-related deaths reported to the Northern Ireland Statistics and Research Agency. In both cases flualprazolam was the cause of death, but toxicity from other substances were also identified.
HSE Advisory Notice December 2019
We are aware that the contents of ‘fake tablets’are changing rapidly. In December 2019, an advisory notice from the HSE National Clinical Lead, Dr Eamon Keenan to all Addiction Series in Ireland.
This notice raised awareness of the changing market and in particular to ‘Nike’ branded tablets that were analysed and found to contain Alprazolam and Tramadol.
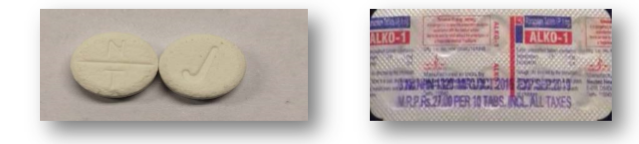
Source:HSE National Social Inclusion Office, 2019
The Benzodiazepine Market and COVID-19
A number of media outlets during the COVID-19 pandemic reported on ‘fake’ tablets circulating and the dangers of purchasing tablets online. As well as this, there were reports of behavioural issues emerging linked to the consumption of ‘fake’ tablets.
Following this, a second notice from the Clinical Lead was issued on the 1st of May 2020 following further concerns in relation to the changing market and combinations of substances that could be emerging. This notice particularly highlighted the risk of overdose if these illicit tablets were consumed as part of a ‘cocktail’ of drugs.
Recently tested substances by the HSE were found to contain Alprazolam and Adinazolam
To reduce the harms associated with the changing benzodiazepine market – the HSE advises the following:
- There could be changes to the market as a result of COVID-19 which increases the risks for people
- You can’t trust the contents of un-prescribed tablets or how you will react
- Changing the type of drug you use or using new drugs increases the risk of overdose
- Start very low and go very slow
- Avoid using more than one drug at a time
This market is constantly evolving and new substances are emerging onto the scene on a regular basis. Services should keep informed of trends and continue to link in with drugs.ie for up to date development and links to reports
Download: Feature I HSE COVID-19 Overdose awareness poster I HSE Advisory Notice to Addiction Services dated May 1st I Drug Watch Factsheet Alprazolam and Etizolam
Sources used in feature:
National Social Inclusion Office (2020), Advisory Notice: New combinations of substances being sold as benzodiazepines to the Irish drug market
Additional information in relation to emerging drug trends and tested substances was obtained from the HSE Social Inclusion records.
Authors: HSE National Social Inclusion Office.
Contact: drugs.ie@hse.ie or Nicki.killeen@hse.ie













